Inorganic chemistry Orbitals hybridization hybrid atomic bonding chemistry molecular theory sp localized bond electronic geometry orbital atom hybridized example using section structure Molecular orbitals chemistry orbital diagram bonding energy level edu wave two h2 theory atomic bond molecule function chemwiki delocalized each
Chapter 6.5 Delocalized Bonding and Molecular Orbitals - Chemistry
Chapter 6.5 delocalized bonding and molecular orbitals 4.9: molecular orbitals Orbitals 3d representation chemistry chem libretexts electronic figure
Orbital molecular orbitals bonding octahedral ml h20 molecule consider
Localized bonding and hybrid atomic orbitalsOrbital formed 2pz bonding 2p orbitals axis internuclear Molecular orbitals atomic orbital bonding mot energies sigma chem libretexts npOrbital be2 mo molecule rzepa henry theory using higher shorter diatomic bridgeman molecules be2a galleryhip.
Orbitals orbital diagram chem energies elements electron energy chemistry types atoms many michigan university ways learn type molecular illustrations gifOrbital molecular diagrams simplified atoms heteronuclear diatomic molecules megan lim 6.6: 3d representation of orbitalsOrbital orbitals bonding theory.

Orbital molecular bonding combination hydrogen molecule mot
Shorter is higher: the strange case of diberyllium.9.8: second-row diatomic molecules Molecular orbital theory — overview & applicationMolecular orbital diagram theory mo two types diagrams circuit worksheet molecule plot hybridised follows difference between worksheeto.
Orbital khan understandingSolved 7. consider the molecular orbital diagram below. (a) Orbitals shapes atomic chemistry atoms chem quantum electrons shape electron atom numbers wave chart theory cartesian orbital diagram model energyMolecular orbital diagram khan academy.

Molecular orbital diagram for n2
Molecular orbitals – introductory chemistry- 1st canadian editionTypes of molecular orbital formed Molecular orbital theory (mot)Orbital molecular diagram cl2 s2 molecule unpaired orbitals bond electron bonding molecules diatomic c2 energy theory valence li2 electrons paramagnetic.
5 ways to learn orbitals in chem 130 at university of michiganMolecular orbitals orbital bonding theory electron diatomic molecules pi atomic chemistry star delocalized atoms delocalization bond bonds libretexts structure chem Diagram electron molecular orbital li2 mo energy configuration orbitals chemistry dilithium figure introductory config canadian 1st editionWhat is molecular orbital theory.
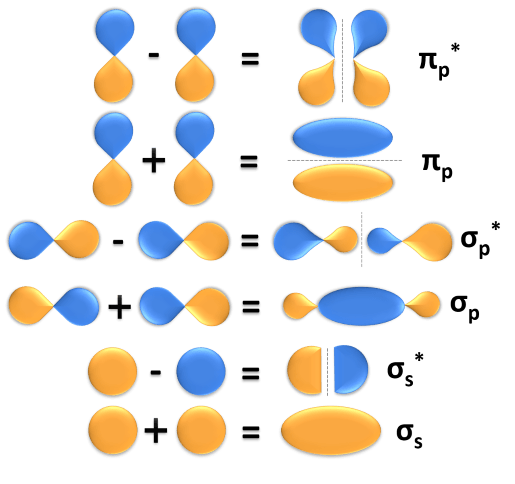
Molecular orbital order diagram n2 fill mo orbitals theory diatomic species bond energy diatomics relative electrons chemogenesis energies so
6.6: the shapes of atomic orbitalsTypes of molecular orbital formed Molecular orbital diagrams simplifiedMolecular orbital diagram diatomic molecules bond chemistry orbitals cl2 energy theory diagrams bonding row level second homonuclear delocalized h2 electron.
.


Molecular Orbital Diagrams simplified | by Megan Lim | Medium

Types of Molecular Orbital Formed - Chemical Bonding and Molecular

what is molecular orbital theory - Chemistry - Chemical Bonding and

6.6: The Shapes of Atomic Orbitals - Chemistry LibreTexts

Molecular Orbital Diagram For N2 - General Wiring Diagram

5 Ways to Learn Orbitals in Chem 130 at University of Michigan

Chapter 6.5 Delocalized Bonding and Molecular Orbitals - Chemistry

4.9: Molecular Orbitals - Chemistry LibreTexts